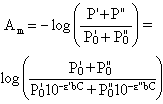|
Deviations from Beer's Law: Effect of Polychromatic Radiation
Theory According to Beer's law, the absorbance A (for a monochromatic radiation beam) of a solution containing an absorbing compound Χ, is proportional to its concentration C, and is given by:
Where ε is the molar absorptivity of X at the particular wavelength, and b is the optical path length. Beer's law is subjected to certain real and apparent deviations. Real deviations are most usually encountered in relatively concentrated solutions of the absorbing compound (>0,01 M). These deviations are due to interactions between the absorbing species and to alterations of the refractive index of the medium. Most common are the apparent deviations. These deviations are due to: (1) chemical reasons arising when the absorbing compound, dissociates, associates, or reacts with a solvent to produce a product having a different absorption spectrum, (2) the presence of stray radiation, and (3) the polychromatic radiation.
Deviations due to polychromatic radiation Strict adherence to Beer's law is observed only with truly monochromatic radiation. Monochromators are used to isolate portions of the output from continuum light sources, hence a truly monochromatic radiation never exists and can only be approximated, i.e. by using a very narrow exit slit on the monochromator. Let's assume that the incident radiation consists of just two wavelengths λ' and λ", with powers P0' and P0". Considering that A = -log(P/P0), then the power of the radiation to come out from (P) the cell for each wavelength would be:
Where ε' and ε" are the molar absorptivities for each wavelength. Therefore the measured absorbance Am will be
The last equation indicates a non-linear relation between Am and C. The proportionality between Am and C is restored only if ε' = ε". The same situation occurs when a radiation consists of many wavelengths. The situation is illustrated in the following figure:
When a polychromatic radiation beam is used, its preferable position (in terms of central wavelength) is on the top of a relatively wide absorption peak (position 1). In this case the proportionality between absorbance (A) and concentration (C) is maintained, because the molar absorptivities are practically the same for all wavelengths. On the other hand, marked deviations are expected when the band is positioned in spectral regions such as the sides of absorption peaks (position 2), where a wide range of molar absorptivity values is expected.. It has been found experimentally that for absorbance measurements at the maximum of narrow peaks, deviations from Beer's law are insignificant, if the effective bandwidth of the incident beam is less than 1/10 of the width of the absorption peak at half height.
Applet With this easy-to-use applet, the deviations from Beer's law due to polychromaticity of the incident radiation beam are easily demonstrated. On the left screen of the applet a hypothetical absorption spectrum consisting of two gaussian peaks (a wide and a narrow one) is depicted. On the right screen the response curve is shown, i.e. the plot of the measured absorbance vs. the concentration, A=f(C), which is normally represented by a straight line passing through the origin. There are only two controls: A scroll-bar for selecting the central wavelength and a second one for adjusting the exit slit of the monochromator (i.e. the width of the incident radiation beam). By moving the central wavelength of incident beam at various positions of the spectrum, one can easily observe the corresponding A=f(C) on the right screen. The red line represents the actual A=f(C) graph, whereas the blue line represents the ideal linear relation, which one would expect if a clearly monochromatic beam was used. Marked adherence to Beer's law is observed by using vary narrow exit slit, or by using the wide band peak wavelength. On the other hand, deviations are observed when wide exit slits are used and particularly when the central wavelength is on the sides of the narrow peak. |